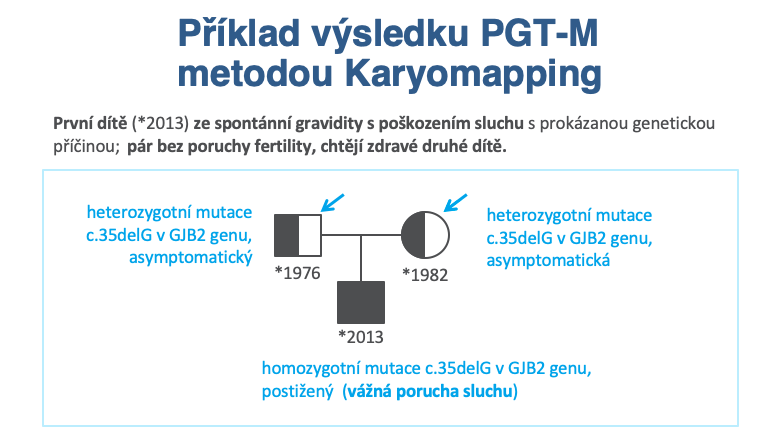
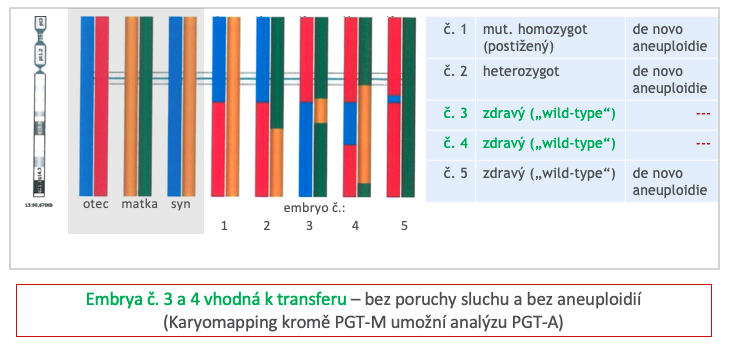
Former name PGD: Preimplantation Genetic Diagnosis for monogenic diseases
Preimplantation genetic testing for monogenic diseases (PGT-M) is a targeted diagnosis of specific severe familial monogenic diseases (autosomal recessive, autosomal dominant, or X-linked), where there is a high risk of giving birth to an affected child. This examination is always conducted based on the indication of a clinical geneticist after evaluating the family history.
INDICATIONS FOR TESTING:
• Carrier of a predisposition (mutation) for a severe genetic disease caused by a defect in a single gene in one or both partners, which is associated with a high risk of giving birth to a child affected by the disease.
TESTING METHODS:
PGT-M is performed on the Karyomapping platform (Illumina), which allows robust testing of monogenic diseases based on indirect diagnosis and also provides an indicative screening for quantitative changes in all chromosomes (PGT-A). We ensure testing for monogenic diseases (PGT-M) in collaboration with contracted laboratories.
TISSUES EXAMINED:
The examination is performed on trophectoderm cells taken on the 5th-6th day of embryo development. Only exceptionally, if for some reason trophectoderm cells cannot be obtained, it may be performed on blastomeres.
COURSE OF THERAPY:
If you decide to undergo therapy with preimplantation testing, its course will be similar to "usual" therapy with IVF-ICSI. The only difference will be that a biopsy will be performed on the embryo on the 5th-6th day of development – that is, the removal of several trophectoderm cells. This sample will then be tested in a genetic laboratory, and the biopsied embryos will be frozen. Once the test results are available, the IVF-ICSI cycle will continue with the transfer of a healthy embryo.